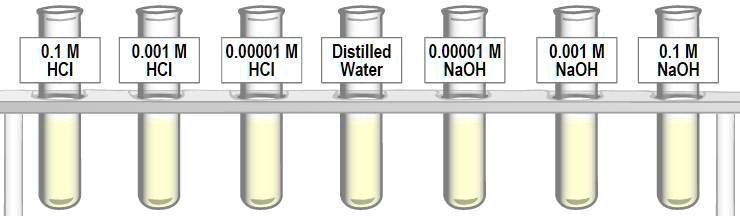
SOLVED: 'What are the pH of these solutions? 0.1 M HCI 0.001 M HCI 0.00001 M HCI Distilled Water 0.00001 M NaOH 0.001 M NaOH 0.1 M NaOH'

SOLVED: pH What is the pH of 0.0M HCI solution? Ans What is the pH of 0.02M HCL solution? Ans 1.7 What is the pH of 0.0[M NaOH solution? ZI 'SUV What

Calculate pH of the following solutions: (i) 0.001M HNO3 (ii) 0.005M H2SO4 (iii) 0.01M KOH (iv) 10^-8M NaOH (v) 0.0008M Ba (OH) 2

SOLVED: The pH of the solution prepared by dilution 1000 times of 0.001 M NaOH solution is: 12 10 A 500 mL of river water contains 0.06 g of mercury: the concentration

Calculate pH for: (a) `0.001 NaOH`, (b) `0.01N Ca(OH)_(2)`, (c ) `0.01M Ca(OH)_(2)`, (d) `10^(-8 - YouTube

Calculate pH of the following solutions: (i) 0.001M HNO3 (ii) 0.005M H2SO4 (iii) 0.01M KOH (iv) 10^-8M NaOH (v) 0.0008M Ba (OH) 2

Calculate pH for: (a) 0.001 NaOH, (b) 0.01N Ca(OH)(2), (c ) 0.01M Ca(OH)(2), (d) 10^(-8)M NaOH, (e ) 10^(2)MNaOH, (f) 0.0008MMg(OH)(2) Assume complete ionisation of each.
Calculate pH for : (a) 0.001 N NaOH, (b) 0.01 N Ca(OH)2 - Sarthaks eConnect | Largest Online Education Community
What is the pH of the resulting solution when the equal volume of 0.1M Noah and 0.01M HCL are mixed? - Quora







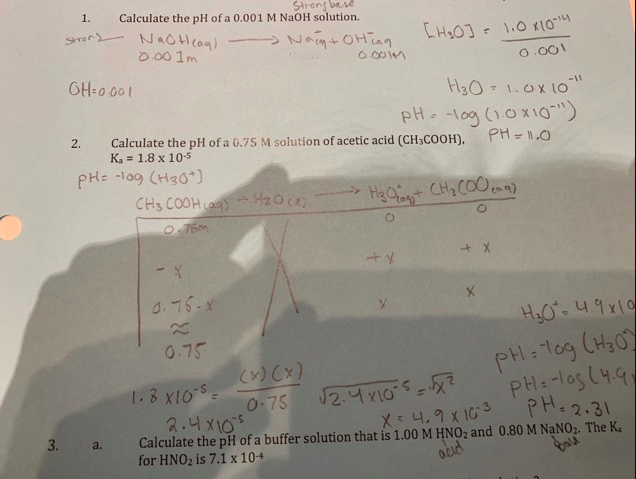

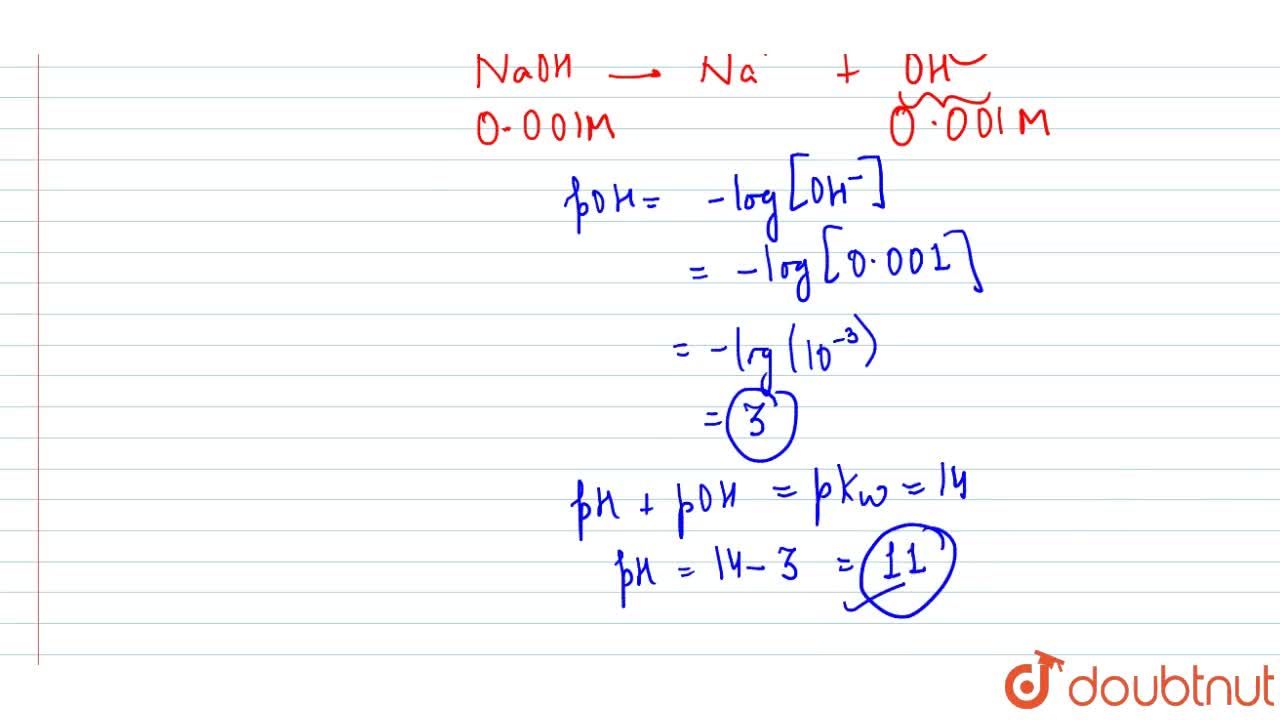




![Solved Calculation of pH, pOH, [H^+] of Acids and Bases | Chegg.com Solved Calculation of pH, pOH, [H^+] of Acids and Bases | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2Fc1f%2Fc1f7b306-69df-4e25-b874-3a1184640a55%2Fimage)
